By John Halamka
This analysis was written by Micky Tripathi and John Halamka.
On Friday March 20, CMS released the Electronic Health Record Incentive Program-Stage 3 and ONC released the 2015 Edition Health Information Technology (Health IT) Certification Criteria, 2015 Edition Base Electronic Health Record (EHR) Definition, and ONC Health IT Certification Program Modifications.
Perhaps the most important statement in the entire 700+ pages is the following from the CMS rule: “Stage 3 of meaningful use is expected to be the final stage and would incorporate portions of the prior stages into its requirements.”
Providers and vendors alike were all hoping for something lean and clean. The CMS Stage 3 rule weighs in at 301 pages, but the ONC Certification rule takes the cake at 431 pages. The JASON Task Force, whose recommendations were unanimously approved by the HIT Standards and Policy Committees, recommended that ONC and CMS make an explicit trade-off: Decrease the breadth and complexity of the MU program, and in return, increase the expectations in a few key areas, such as interoperability. The CMS MU Stage 3 rule, for the most part, has adopted this philosophy. Unfortunately, the same can’t be said for the ONC Certification rule.
We provide a brief synopsis of the MU and Certification Rules below, followed by our analysis of these proposals.
CMS Stage 3 MU Rule Synposis
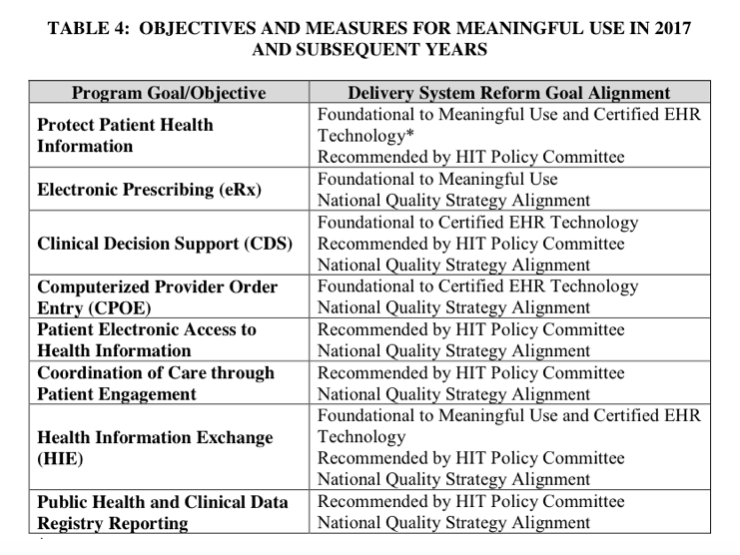
The CMS Meaningful Use Rule is focused and narrowed to 8 objectives.
There is some fine-print though. Contained within many of the objectives are multiple measures. Depending on which options one chooses, and whether you are a provider or a hospital, the total number of MU measures could range from 15 to 20, and that’s NOT INCLUDING the Clinical Quality Measures, which have always been like a MU menu all of their own, and which are now going to be determined through a different process and won’t be defined until later in 2015.
Here is a synopsis of the MU Stage 3 requirements:
Provider-facing EHR functions:
*ePrescribing: The thresholds have increased to 80% for EPs and 25% for EHs, but overall this is just asking for more of the same. Of note is that controlled substance prescriptions can now be optionally included in states where it is allowed electronically.
*Clinical decision support: There are 2 measures: 1) implement 5 CDS interventions tied to 4 quality measures; and 2) turn on drug-drug and drug-allergy interaction alerts for the entire EHR reporting period. This is aligned with the past trajectory from earlier stages.
*CPOE: There are 3 measures: use CPOE on at least 80% of medication orders, 60% of lab orders, and 60% of diagnostic imaging orders. CMS has given a little flexibility here by now counting entry by “scribes” (personnel with at least a medical assistant credential), excluding standing orders, and including a broader array of imaging …read more
Source:: http://geekdoctor.blogspot.com/2015/03/the-cms-and-onc-nprms_24.html